Volume 1 | Issue 1 |Apr – June 2016 | Page 15-20|H S Chhabra[1], PK Kartik Yelamarthy[1].
Authors :H S Chhabra[1], PK Kartik Yelamarthy[1]
[1] Indian Spinal Injuries Centre, Sector C, Vasant Kunj,
New Delhi, India.
Address of Correspondence
Dr. H S Chhabra
Indian Spinal Injuries Centre, Sector C, Vasant Kunj, New Delhi, India.
Email Id:-dryelamarthykarthik@gmail.com
Abstract
Adult spinal deformity may occur as the result of a number of conditions and patients may present with a heterogeneous group of symptoms. These include deformities in all 3 planes (coronal, sagital and axial) i.e scoliosis, kyphosis and rotational deformities. Clinical presentation varies and differs from pediatric deformities in that patients present more often with axial back pain and neurogenic claudication rather than a cosmetic deformity. Indications for treatment include pain, neurogenic symptoms, and progressive deformity. Larger deviations in the anterior, posterior or lateral plane will require greater energy use to maintain a standing position. Finally, progression outside of the “stable cone of economy” results in a loss of postural control and the need for external supports. The aim of this article is to analyze the role of osteoporosis in the progression of adult spinal deformity .We conducted a review of literature from Medline and searched for articles related to adult spinal deformities and osteoporosis. Osteoporosis along with progressive and asymmetric degeneration of the disc and facet joints has a role in progression of these deformities. Osteoporosis may also impact surgical options and can significantly impact the operative plan.
Keywords: Adult spinal deformity, osteoporosis, disc degeneration, deformity progression.
Introduction
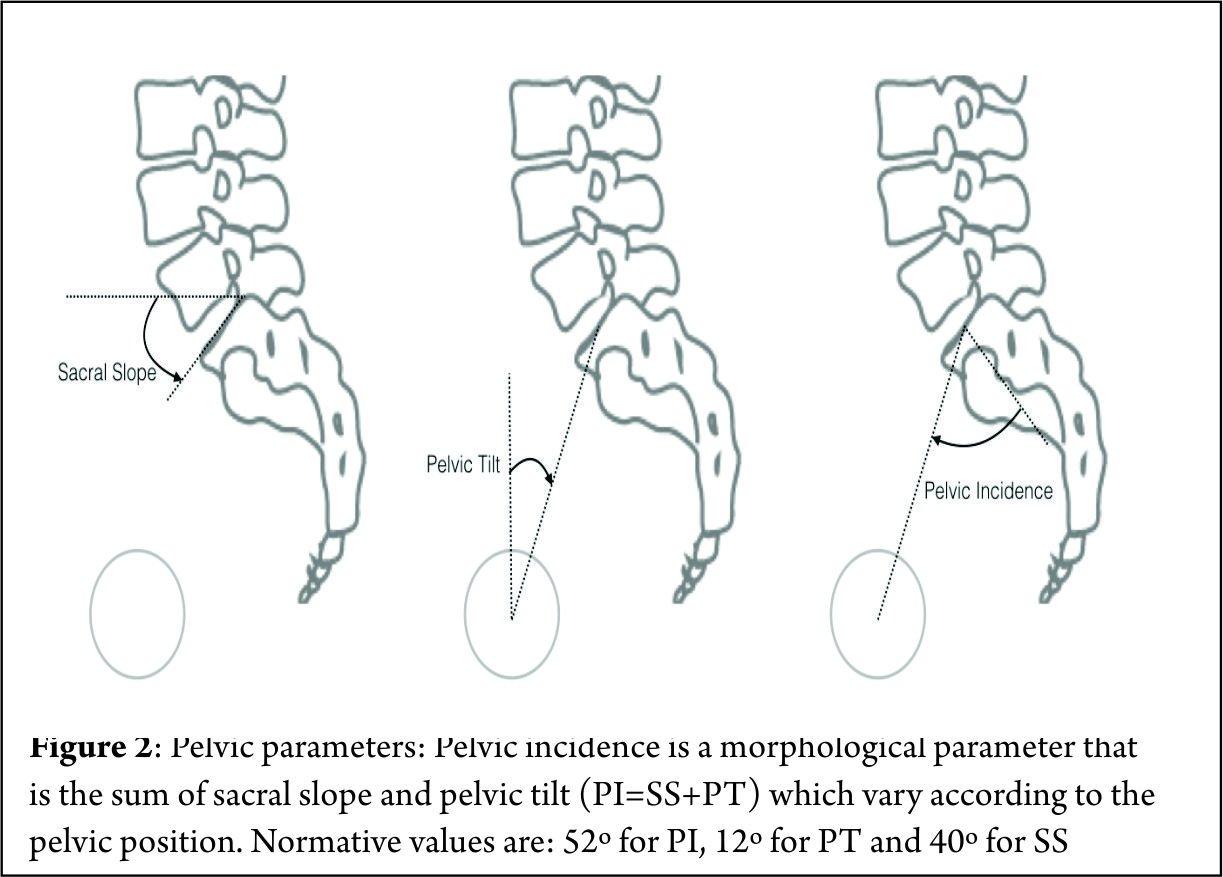
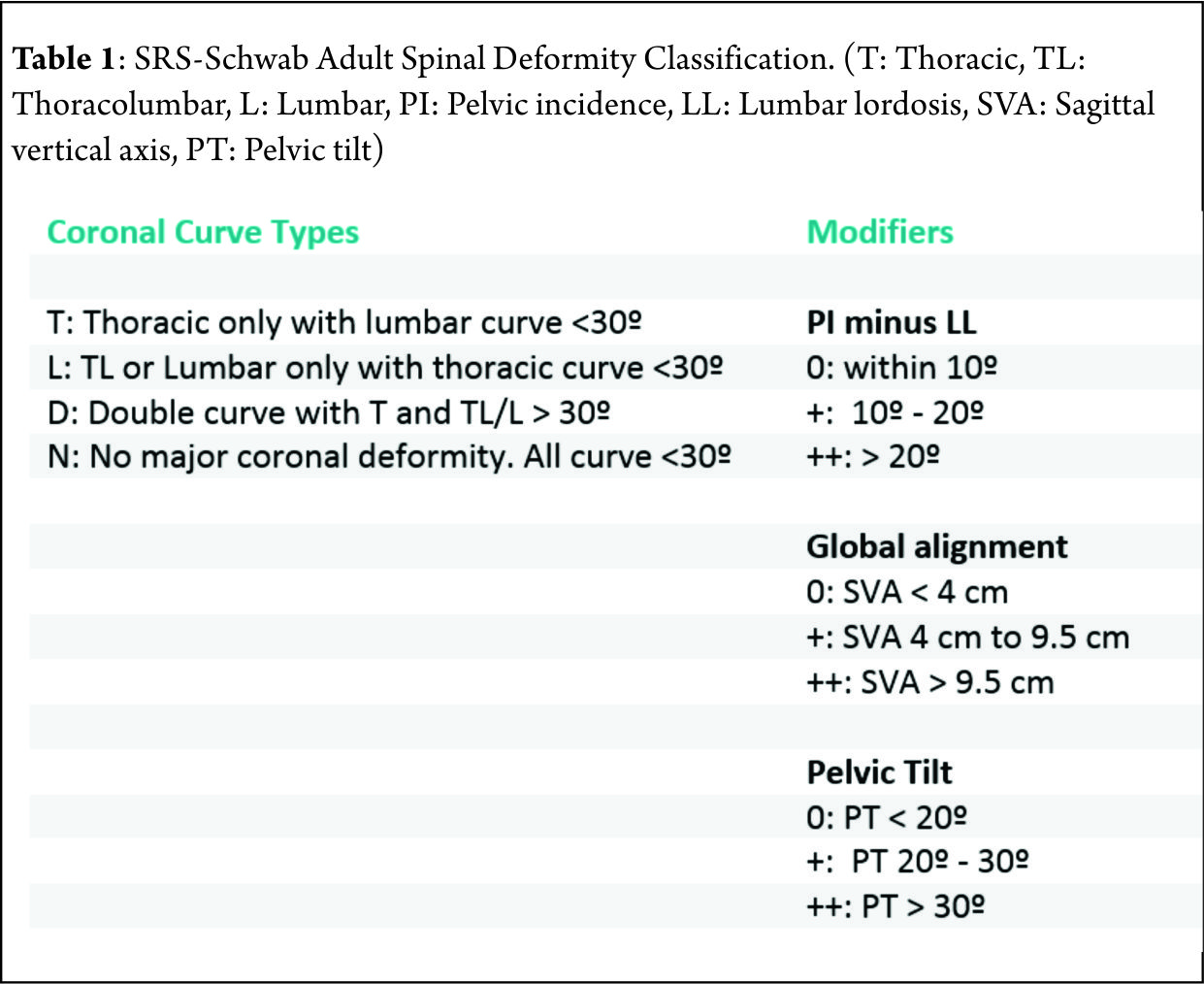
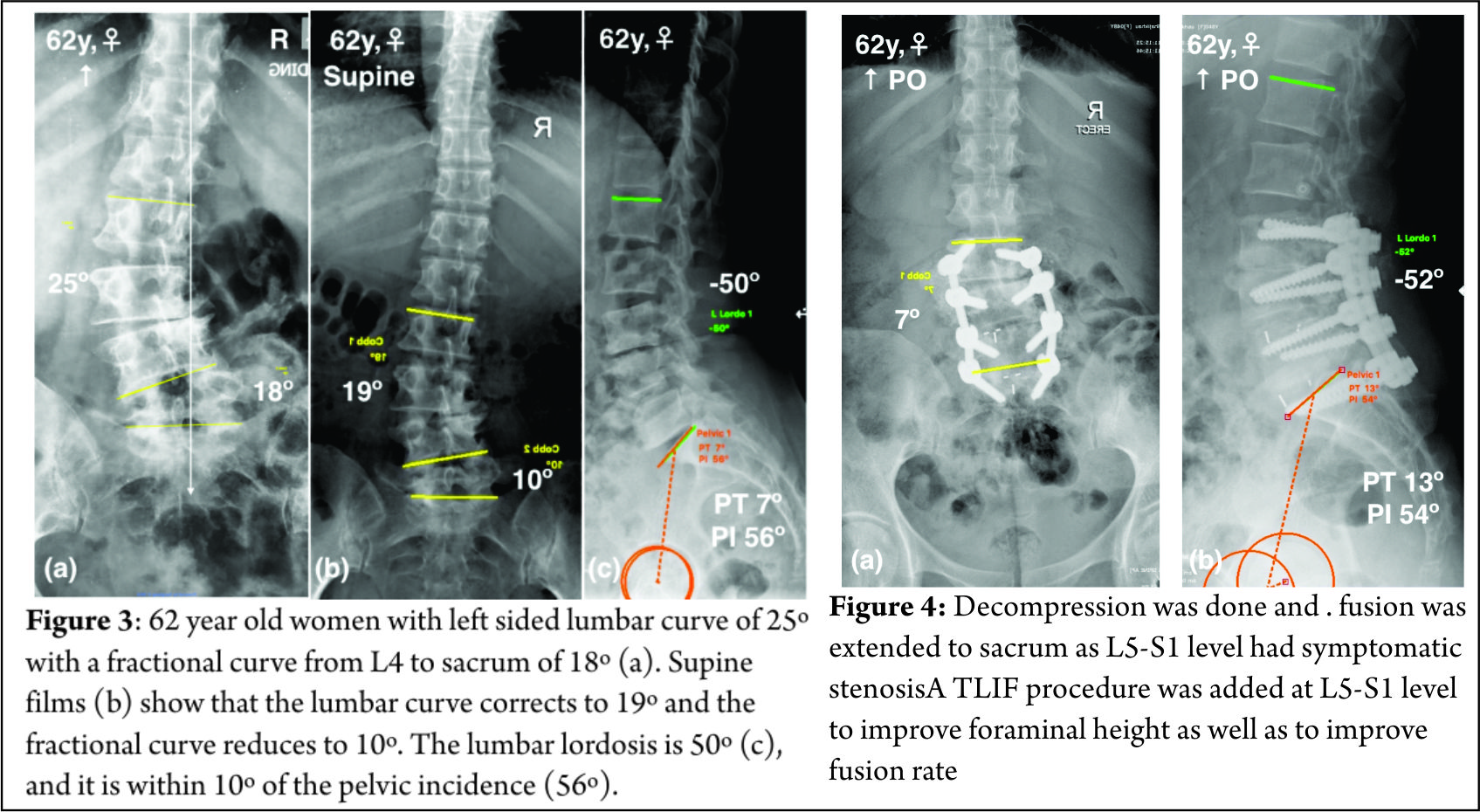
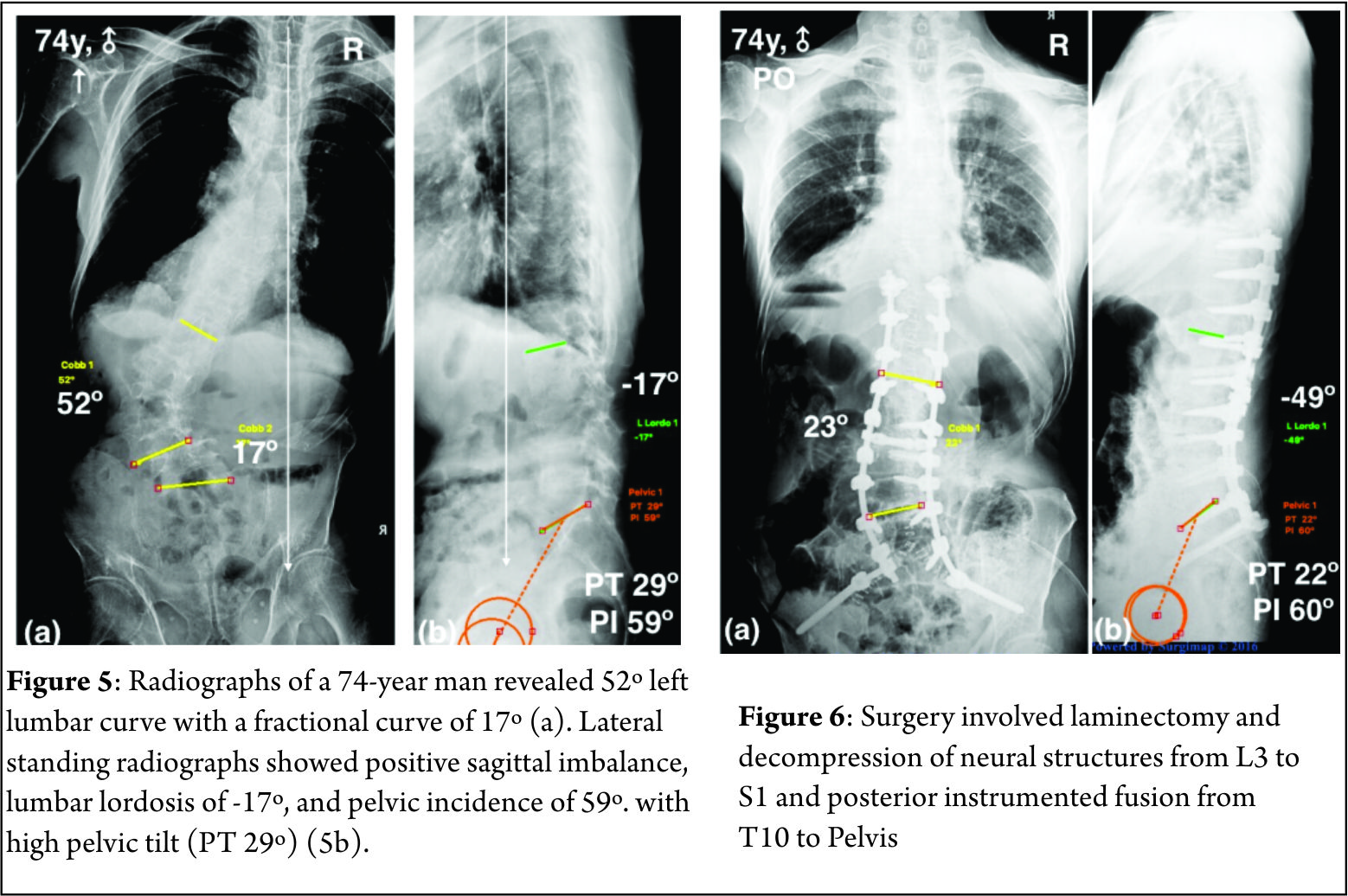
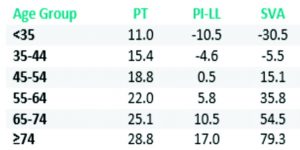
Spinal deformity is defined as a curvature in the spine where the alignment is outside of defined normal limits. Adult spinal deformity is one of the most challenging spinal disorders and by definition describes a complex spectrum of spinal deformities that present in adulthood including adult scoliosis, sagittal and coronal imbalance, and iatrogenic deformity, with or without spinal stenosis [1].Adult spinal deformity may occur as a result of a number of conditions, each of which ultimately lead to an imbalance of the structural support of the spinal column. Abnormal curvature may occur in the sagittal plane (kyphosis, lordosis) or in the coronal plane (scoliosis) causing imbalance in both planes . The magnitude of the curvature of the spine is measured using Cobb angle measurements. Axial plane deformity is measured by degrees of rotation from the frontal or sagittal plane [2]. Clinical presentation of adult spinal deformity varies greatly from minimal or no symptoms to severe pain with disability [3]. A majority of patients remain asymptomatic with radiographic findings alone. However, when patients begin to complain of symptoms, these may vary from mild back pain without radiculopathy to severe back pain with neurogenic claudication, radiculopathy, and walking intolerance.4 A complete patient assessment requires not only appropriate imaging studies but a complete history and physical exam. An adult deformity classification has been established by Schwab et al and applies radiographic parameters of disability[5]. As presented in Table 1. The presented adult deformity classification has a significant impact on surgical rates and operative strategy (approach, fusion to sacrum, and use of osteotomies). Through continued investigation, further refinement of the classification and formation of effective treatment algorithms are certain to emerge to guide the care of adults suffering from spinal deformity [5].
Scoliosis: A scoliosis is diagnosed in adult patients when it occurs or becomes relevant after skeletal maturity with a Cobb angle of more than 10 degrees in the frontal plain film [6,7].
Type 1: Primary degenerative scoliosis (‘‘de novo’’ form), mostly located in the thoracolumbar or lumbar spine
Type 2: Progressive idiopathic scoliosis in adult life of the thoracic, thoracolumbar, and/or lumbar spine
Type 3: Secondary degenerative scoliosis.
(a) Scoliosis following idiopathic or other forms of scoliosis or occurring in the context of a pelvic obliquity due to a leg length discrepancy, hip pathology or a lumbosacral transitional anomaly, mostly located in the thoracolumbar, lumbar or lumbosacral spine .
(b) Scoliosis secondary to metabolic bone disease (mostly osteoporosis) combined with asymmetric arthritic disease and/or vertebral fractures
Clinically, the most prominent groups are secondary (type 3) and primary (type 1)degenerative adult scoliosis. In elderly patients, all the three forms may be aggravated by osteoporosis [8,9,10].
Sagittal Plane Deformity: Age-related postural hyperkyphosis is an exaggerated anterior curvature of the thoracic spine, sometimes referred to as Dowager’s hump or gibbous deformity. This condition impairs mobility [22] and increases the risk of falls [23] and fractures [24]. Several types of postural deformities exist according to the number, severity, and location of vertebral fractures(upper or middle thoracic, thoracolumbar, or lumbar).Satoh et al. classified osteoporotic postural deformities into the following five groups based on changes of the physiological thoracic and lumbar curvature:[11]
1) normal posture without apparent change in spinal curve;
2) round back with increased thoracic kyphosis and normal lumbar lordosis;
3) hollow round back with increased thoracic kyphosis and lumbar lordosis;
4) whole kyphosis with extensive kyphosis from thoracic to lumbar spine; and
5) lower acute kyphosis with localized lumbar kyphosis with straight thoracic spine
Iatrogenic Spinal Deformity
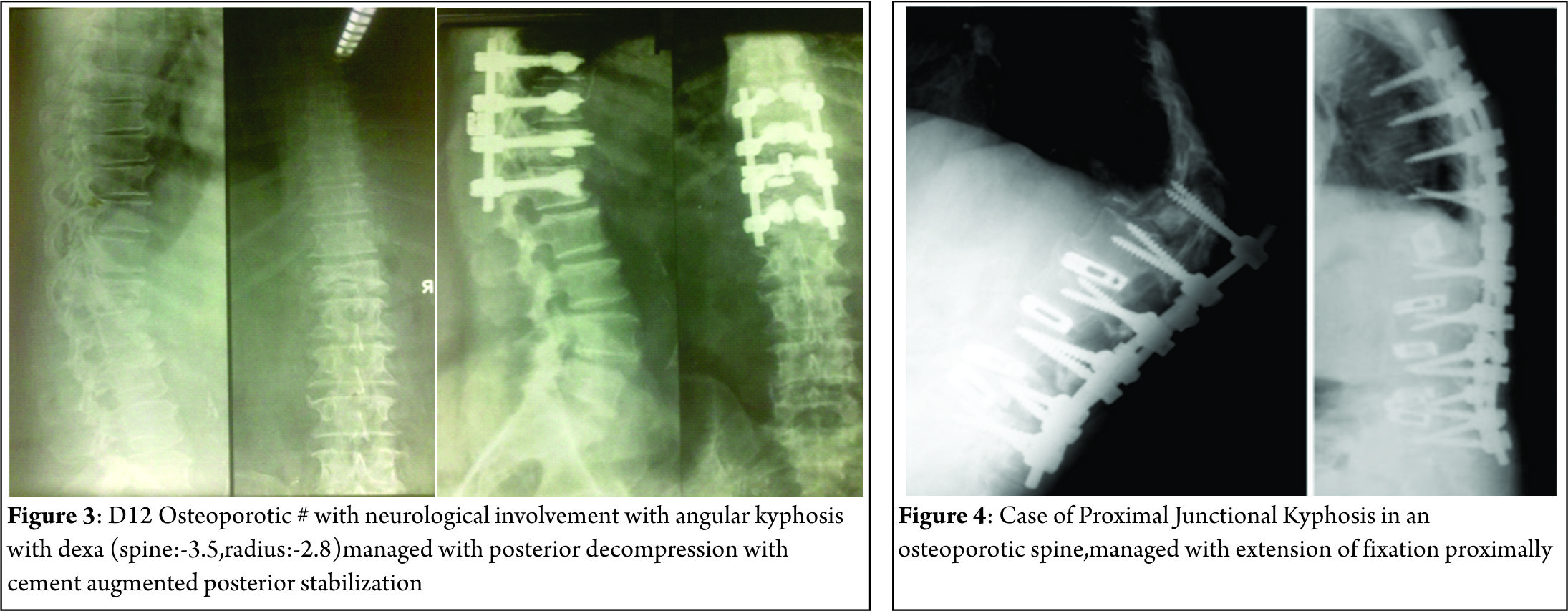
Iatrogenic spinal deformities can cause either sagittal or coronal imbalance. They consist of flat back syndrome, post-laminectomy kyphosis and proximal junctional kyphosis(PJK).With the development of posterior segmental stabilization, the rates of flat back syndrome decreased [12,13,14,15,16]. A pathology known as junctional kyphosis also commonly calls for the use of instrumentation above the level of fusion in the thoraco-lumbar or cervico-thoracal vertebrae . Fusions that end at the level of the 7th and 8th thoracic vertebrae are known as the apex of thoracic kyphosis and may lead to junctional kyphosis. Post-laminectomy kyphosis is mostly seen after multi-level laminectomy procedures, especially in the cervical region and in cases of facetectomies with facet capsule destruction [17]. After cervical laminectomies in pediatric patients with incomplete bone development, post-laminectomy kyphosis is seen more commonly than in adult patients [17,18]. The rate of post-laminectomy kyphosis may become much greater in the population of pediatric patients with malignant intramedullary pathologies following radiotherapy treatment [17,19].
Natural History
Adult Scoliosis:
Idiopathic curves:(type 2 curves)[20]:
Lumbar curves of more than 300 degrees with apical vertebral rotation of more than 30% progressed the most. Right sided lumbar curves tended to progress twice as much as left lumbar curves. Also, marked vertebral rotation combined with translational shift (lateral olisthesis) was associated with significant curve progression. The thoracolumbar curve pattern manifested the most pronounced amount of apical vertebral rotation. The incidence of translatory shifts increased with time. Combined curves tended to balance with age, although lumbar part tended to progress more than the thoracic counter part. Weinstein and Ponsetti noted greater progression in lumbar curve if L5 was not well seated over the sacrum and apical vertebral rotation was more than 33% [48].
De novo curves (type 1 curves)[20]:
Robin et al analysed 554 individuals longitudinally for 7 to 13 years (315 women and 239 men, age range 50 to 84 yrs). 179 had curves exceeding 100. Fifty five (10%) developed denovo scoliosis during this period. Left sided curves were common in women. Sex ratio (F:M) increased with curve size. Rotatory olisthesis was found in 34% of patients, most common at L3-4 and L4-5 levels. As per a prospective study by Korovessis et al [21], risk factors directly related to curve progression were lateral olisthesis at the apical vertebra, a high Harrington factor (Cobb angle divided by number of vertebrae included in the curve) and the disc index.
Sagittal Plane Deformity:
The natural history of hyperkyphosis is not firmly established. Hyperkyphosis may develop from either muscle weakness and degenerative disc disease, leading to vertebral fractures and worsening hyperkyphosis, or from initial vertebral fractures that precipitate its development
Cause And Progression Of Adult Deformity (scoliosis, sagittal deformity, iatrogenic deformity)
Scoliosis:
The clinical syndrome of spinal osteoporosis is characterized by the occurrence of non-traumaticvertebral fractures and a disproportionately large amount of loss of trabecular bone necessary for the maintenance of vertebral strength [25,26].
Significant correlations exist between the bone mineral content, the compressive strength of the vertebrae [27,27,29], vertebral fractures, hyperkyphosis, and back pain. What remains unresolved is the role played by decreased vertebral bone mineral content in the development and progression of scoliosis in the adult. Shands and Eisberg noted a higher incidence of scoliosis in persons over 60years of age. Later, Vanderpool et al [30] reported scoliosis in 6%of persons over 50 years, and 36%more in those with osteoporosis. Those authors noted that scoliosis can arise in the elderly and is etiologically related to the higher incidence of metabolic bone disease. In an epidemiological study of routine anteropostenor (AP) chest and recumbent roentgenograms of the lower thoracic and lumbar spine, Robin et a1 .concluded that there was no basis for assuming a causal relationship between scoliosis and osteoporosis .But several theories could explain the association between fractures and scoliosis. Fractures may cause scoliosis, or, conversely scoliosis may cause fractures. Alternatively, scoliosis and fractures may be manifestations of the same underlying condition so that they would be expected to occur together frequently. The posterior spinal elements, facet joints. and ligamentous structures are not disrupted in osteoporotic compression fractures and may provide a fixed axis for sagittal and coronal spinal deformities. Yet this explanation does not account for the rotatory component of the deformity seen in osteoporotic patients. Mechanical factors that increase the forces applied to a vertebra increase the likelihood of that vertebra mechanically failing. Theoretically, a collagen abnormality may be responsible for osteoporosis and so called idiopathic scoliosis. Thus, several independent lines of evidence support the finding of a high concordance between osteoporosis and scoliosis. Fractures in osteoporotic or scoliotic patients would result from routine loading of an inherently weak spinal connective tissue [31].
The asymmetric degeneration of the disc and/or the facet joints leads to an asymmetric loading of the spinal segment and consequently of a whole spinal area. This again leads to an asymmetric deformity. Such a deformity again triggers asymmetric degeneration and induces asymmetric loading, creating a vicious cycle and enhancing curve progression. The destruction of structural spinal elements like discs, facet joints, and joint capsules responsible for stability leads to uni- or multi-segmental, multi-directional instability and can manifest as spondylolisthesis or translational or rotary olisthesis. The biological reaction is the formation of osteophytes at facet joint and vertebral end plates contributing to increasing narrowing of the spinal canal with facetjoint and ligamentum flavum hypertrophy and calcification. Effective narrowing of the spinal canal caliber causes central and lateral recess spinal stenosis [32,33]. Instability and collapse of the disc height leads to foraminal stenosis, with radicular pain or neurogenic claudication-type pain.
The asymmetric loading, coupled with degeneration, triggers a vicious cycle enhancing curve progression. This is fueled by common metabolic bone disorders like osteoporosis especially in post-menopause female patients leading to further asymmetric deformation and collapse in the weakened osteoporotic vertebra with subsequent curve progression[34].

Sagittal Plane Deformity:
Sagittal postural deformities begin with localized kyphosis due to either fracture or asymptomatic insidious collapse at the thoracic and/or thoracolumbar spine . This increased kyphosis (round back) can be readily compensated by increasing lumbar lordosis, resulting in the formation of the hollow round back [35]. Compensated upper thoracic lordosis for wedged vertebral fractures at the thoracolumbar junction results in lower acute kyphosis .If progressing round back cannot be compensated by lumbar lordosis, kyphosis extends down to the lumbar region, resulting in whole kyphosis. Therefore, whole kyphosis cannot be compensated by other spinal segments. Because lumbar kyphosis is thought to be related to weakness of the spinal extensors [36], whole kyphosis usually forces the patient to use a cane while standing and walking [35]. These uncompensated conditions seemed to contribute impairment of all the domain scores in the whole kyphosis group, especially in the activities of daily living domain score, resulting in a significant reduction in the total quality of life score compared with other postural deformities.
Iatrogenic Deformity:
Risk factors for PJK included age at operation, low bone mineral density, shorter fusion constructs, upper instrumented vertebrae below L2, and inadequate restoration of global sagittal balance. Osteopenia/osteoporosis has been established as a significant risk factor for proximal junctional kyphosis. Both symptomatic and asymptomatic compression fractures that kyphose the spine are not uncommon in the elderly. In addition, the elderly tend to have more kyphosis in their thoracic spine. For this reason, longer instrumented fusions that span the entire thoracic spine are often needed [37]
Implications of Deformity Progession:
In the domain of spinal surgery, it is useful to recall important concepts that can serve as a foundation to understanding and treating deformity. Optimal alignment of bone structures and joints is critical for the efficient function of the musculoskeletal system. Furthermore, a complex interaction of the neurologic system and muscular recruitment is necessary for ergonomic balance and deliberate displacement of the human body. Therefore, it is important to consider that ideal spinal alignment allows an individual to assume standing posture with minimal muscular energy expenditure. Physiologic curvatures of the spine in the sagittal plane, the straight spine in the coronal plane, balanced tension of the spinal ligaments, and activation of intrinsic anterior and posterior musculature should permit extended pain free erect position. This concept is reflected in the “Cone of Economy” principle conceptualized by Jean Dubousset [46] (Figure 1). Within the center of the cone, the individual may remain in an ergonomically favorable erect position. However, larger deviations in the anterior posterior
or lateral plane will require greater energy use to maintain a standing position. Finally, progression outside of the “stable cone” results in a loss of postural control and the need for external supports.

In the setting of adult spinal deformity (ASD), structural or iatrogenic modifications to spinal alignment should be considered . Spinal malalignment in ASD challenges balance mechanisms used for maintenance of an upright posture to achieve the basic human needs of preserving level visual gaze and retaining the head over the pelvis. Progressive severity in skeletal malalignment might result in greater recruitment in muscular effort and greater energy expenditure to maintain the erect posture as well as use of compensatory mechanisms. Spinal malalignment to the extremes of the “Cone of Economy” leads to extreme muscular demand, fatigue, and significant pain as well as disability. Once a spinal deformity has reached
the level of marked loss in function and quality of life, surgical intervention is often recommended and requested [46,47]
Influence of Osteoporosis on Management of Adult Deformity
The surgical treatment is complicated by the weak bone where implants are more difficult to be anchored and fixed, making the instrumented fusion prone to instrumentation-related complications. Trabecular bone is predominantly affected by osteoporosis, and because the pedicle screw has cortical contact limited to the pedicle isthmus, a “windshield wiper” mode of failure typically leads to screw loosening [38]. Therefore, fixation strategies for osteoporotic bone are targeted either toward taking advantage of the relatively stronger cortical bone [39] or toward augmenting the fixation of a pedicle screw within the existing trabecular bone [40]. It should be recognized, however, that when sclerosis is associated with degeneration in patients with adult scoliosis, the local bone mineral density may be significantly increased, limiting the local effects of the systemic osteoporosis. Various methods have been used for treatment of the osteoporotic patient, including sublaminar wires and pediculolaminar fixation [41], both of which take advantage of cortical bone composition of the posterior spinal lamina. In addition, techniques to improve the fixation of pedicle screws within osteoporotic trabecular bone have also been developed including polymethylmethacrylate cement augmentation of pedicle screws [42]. Calcium sulfate paste may also be used, which has the theoretical advantage of becoming replaced by bone over time 43. Other alternatives have been investigated, including conical screws, hydroxyapatite-coated screws, and expandable screws. There is no consensus on the optimal screw diameter, length, or shape for fixation in osteoporotic bone. It has been demonstrated that with high insertional torque, the screw-strength is improved [44]. This may be attained by undertapping (or not tapping) the screw trajectory. Larger-diameter screws may offer increased contact with the cortical bone of the pedicle and, thus, increase insertional torque. However, this may potentially increase the risk of pedicle fracture particularly in this population. Longer screw length also can increase screw pull-out strength, particularly when there is “bicortical” purchase with the distal end of the screw passing through the anterior vertebral cortex. However, bicortical purchase increases the possibility of injury to abdominal or vascular structures and therefore is usually limited to the sacral region. An additional strategy is to use multiple points of fixation with a numerous pedicle screw construct thus providing for the spread and distribution of contact forces. Care should be taken to preserve the supraspinous ligament, intraspinous ligament, and ligamentum flavum between the rostral fused level and the adjacent segment as well as throughout the construct where possible. This may possibly prevent the development of junctional deformity and instability because it serves as a segment of high posterior tension. With extension of the fusion to the sacrum, utilization of multiple and bicortical screw fixation in addition to consideration of anterior column support at L5–S1 and/or iliac fixation should be considered. Larger diameters and increased lengths to 70 or 80 mm improve iliac screw pull-out strength. This improved caudal fixation in deformity patients has been found to be persistent in patients with a minimum of 5 years of follow-up [45]. In summary, several of the central tenets of spinal reconstruction are particularly important in the adult deformity patient population with poor bone quality. Appropriate balance reduces junctional forces, which diminishes the risk of both instrumentation failure and adjacent vertebral fractures. The surgeon should thus endeavor to balance the rostral and caudal ends of the construct. In addition, a meticulous fusion procedure, augmented with appropriate bone graft or bone graft substitutes, is especially important. This will support the development of a rapid and solid fusion such that long-term spinal stability will be ensured, relieving the requirements put upon the reconstruction instrumentation and its relatively poor interface with the osteoporotic bone.
Conclusions
Adult spinal deformity may occur as a result of a number of
conditions, each of which ultimately lead to an imbalance of the
structural support of the spinal column. These deformities can
occur in all 3 planes. The asymmetric loading, coupled with
degeneration is fuelled by osteoporosis in the progression of
adult spinal deformities. Osteoporosis also plays a role in
deciding the instrumentation to be used in management of these deformities.
References
1. Birknes JK, White AP, Albert TJ, Shaffrey CI, Harrop JS. Adult degenerative scoliosis: a review. Neurosurgery 2008;63(3, Suppl): 94–103
2. Curr Rev Musculoskelet Med. 2011 Dec; 4(4): 159–167
3. Schwab F, Lafage V, Farcy JP, et al. Surgical rates and operative outcome analysis in thoracolumbar and lumbar major adult scoliosis: application of the new adult deformity classification. Spine 2007;32:2723–2730
4. Sengupta K. Adult spinal deformity. In: Rao RD, Smuck M, eds. Orthopaedic Knowledge Update: Spine, 4th ed. Rosemont, IL: American Academy of Orthopaedic Surgeons; 2012:349–367
5. Schwab F, Lafage V, Farcy JP, Bridwell K, Glassman S, Ondra S, Lowe T, Shainline M. Surgical rates and operative outcome analysis in thoracolumbar and lumbar major adult scoliosis: application of the new adult deformity classification. Spine (Phila Pa 1976). 2007 Nov 15;32(24):2723-30.
6. Schwab F, el-Fegoun AB, Gamez L, Goodman H, Farcy JP. A lumbar classification of scoliosis in the adult patient: preliminary approach. Spine (Phila Pa 1976). 2005 Jul 15;30(14):1670-3.
7. Aebi M. The adult scoliosis. Eur Spine J. 2005 Dec;14(10):925-48
8. Grubb SA, Lipscomb HJ. Diagnostic findings in painful adult scoliosis. Spine (Phila Pa 1976). 1992 May;17(5):518-27.
9. Healey JH, Lane JM. Structural scoliosis in osteoporotic women. Clin Orthop Relat Res. 1985 May;(195):216-23
10. Velis KP, Healey JH, Schneider R. Osteoporosis in unstable adult scoliosis. Clin Orthop Relat Res. 1988 Dec;(237):132-41.
11. Satoh K, Kasama F, Itoi E, et al. Clinical features of spinal osteoporosis: spinal deformity and pertinent back pain. Contemp Orthop. 1988;16:23–30
12. Bridwell KH, Betz R, Capelli AM, Huss G, Harvey C. Sagittal plane analysis in idiopathic scoliosis patients treated with Cotrel-Dubousset instrumentation. Spine (Phila Pa 1976). 1990 Sep;15(9):921-6.
13. Lenke LG, Bridwell KH, Baldus C, Blanke K, Schoenecker PL. Ability of Cotrel-Dubousset instrumentation to preserve distal lumbar motion segments in adolescent idiopathic scoliosis. J Spinal Disord. 1993 Aug;6(4):339-50.
14. Lenke LG, Bridwell KH, Baldus C, Blanke K, Schoenecker PL. Cotrel-Dubousset instrumentation for adolescent idiopathic scoliosis. J Bone Joint Surg Am. 1992 Aug;74(7):1056-67
15. Takahashi S, Delécrin J, Passuti N. Changes in the unfused lumbar spine in patients with idiopathic scoliosis. A 5- to 9-year assessment after cotrel-dubousset instrumentation. Spine (Phila Pa 1976). 1997 Mar 1;22(5):517-23
16. Wiggins WC, Ondra SL, Shaffray CI. Management of flat-back syndrome. Neurological Focus 2003; 15(3): 1-9.
17. Deutsch H, Haid RW, Rodts GE, Mummaneni PV. Postlaminectomy cervical deformity. Neurosurg Focus 2003;15(3): E5
18. Katsumi Y, Honma T, Nakamura T: Analysis of cervical instability resulting from laminectomies for removal of spinal cord tumor. Spine 1989;14:1171–1176.
19. Yeh JS, Sgouros S, Walsh AR, Hockley AD. Spinal sagittal malalignment following surgery for primary intramedullary tumours in children. Pediatr Neurosurg. 2001 Dec;35(6):318-24
20. The Lumbar Spine edited by Harry N. Herkowitz, International Society for Study of the Lumbar Spine
21. Korovessis P, Piperos G, Sidiropoulos P, Dimas A. Adult idiopathic lumbar scoliosis. A formula for prediction of progression and review of the literature. Spine (Phila Pa 1976). 1994 Sep 1;19(17):1926-32.
22. Kado DM, Huang MH, Barrett-Connor E, Greendale GA. Hyperkyphotic posture and poor
physical functional ability in older community-dwelling men and women: the Rancho Bernardo
study. J Gerontol A Biol Sci Med Sci 2005;60:633–637.
23. Kado DM, Huang MH, Nguyen CB, Barrett-Connor E, Greendale GA. Hyperkyphotic posture and risk of injurious falls in older persons: the Rancho Bernardo Study. J Gerontol A Biol Sci Med Sci 2007;62:652–657
24. Huang MH, Barrett-Connor E, Greendale GA, Kado DM. Hyperkyphotic posture and risk of future osteoporotic fractures: the Rancho Bernardo study. J Bone Miner Res 2006;21:419–423
25. Mazess RB. Measurement of skeletal status by noninvasive methods. Calcif Tissue Int. 1979 Oct 31;28(2):89-92.
26. Nordin BE. Clinical significance and pathogenesis of osteoporosis. Br Med J. 1971 Mar 13;1(5749):571-6.
27. Hansson T, Roos B. Microcalluses of the trabeculae in lumbar vertebrae and their relation to the bone mineral content. Spine (Phila Pa 1976). 1981 Jul-Aug;6(4):375-80..
28. Hansson T, Roos B, Nachemson A. The bone mineral content and ultimate compressive strength of lumbar vertebrae. Spine (Phila Pa 1976). 1980 Jan-Feb;5(1):46-55..
29. Hansson T, Roos B. The relation between bone mineral content, experimental compression fractures, and disc degeneration in lumbar vertebrae. Spine (Phila Pa 1976). 1981 Mar-Apr;6(2):147-53.
30. Vanderpool DW, James JI, Wynne-Davies R. Scoliosis in the elderly. J Bone Joint Surg Am. 1969 Apr;51(3):446-55
31. Healey JH, Lane JM. Structural scoliosis in osteoporotic women. Clin Orthop Relat Res. 1985 May;(195):216-23
32.Benner B, Ehni G. Degenerative lumbar scoliosis. Spine 1979; 4:548
33. Ploumis A, Transfledt EE, Denis F. Degenerative lumbar scoliosis associated with spinal stenosis. Spine J. 2007 Jul-Aug;7(4):428-36.
34. Kotwal S, Pumberger M, Hughes A, Girardi F. Degenerative scoliosis: a review. HSS J. 2011 Oct;7(3):257-64.
35. Satoh K, Kasama F, Itoi E et al. Clinical features of spinal osteoporosis: spinal deformity and pertinent back pain. Contemp Orthop 1988;(16):23–30
36. Takemitsu Y, Harada Y, Iwahara T, Miyamoto M, Miyatake Y. Lumbar degenerative kyphosis. Clinical, radiological and epidemiological studies. Spine (Phila Pa 1976). 1988 Nov;13(11):1317-26.
37. Lau D, Clark AJ, Scheer JK, Daubs MD, Coe JD, Paonessa KJ, LaGrone MO, Kasten MD, Amaral RA, Trobisch PD, Lee JH, Fabris-Monterumici D, Anand N, Cree AK, Hart RA, Hey LA, Ames CP; SRS Adult Spinal Deformity Committee. Proximal junctional kyphosis and failure after spinal deformity surgery: a systematic review of the literature as a background to classification development. Spine (Phila Pa 1976). 2014 Dec 1;39(25):2093-102.
38. Law M, Tencer AF, Anderson PA. Caudo-cephalad loading of pedicle screws: mechanisms of loosening and methods of augmentation. Spine (Phila Pa 1976). 1993 Dec;18(16):2438-43.
39. Coe JD, Warden KE, Herzig MA, McAfee PC. Influence of bone mineral density on the fixation of thoracolumbar implants. A comparative study of transpedicular screws, laminar hooks, and spinous process wires. Spine (Phila Pa 1976). 1990 Sep;15(9):902-7.
40. Tan JS, Kwon BK, Dvorak MF, Fisher CG, Oxland TR. Pedicle screw motion in the osteoporotic spine after augmentation with laminar hooks, sublaminar wires, or calcium phosphate cement: a comparative analysis. Spine (Phila Pa 1976). 2004 Aug 15;29(16):1723-30.
41. Hilibrand AS, Moore DC, Graziano GP. The role of pediculolaminar fixation in compromised pedicle bone. Spine (Phila Pa 1976). 1996 Feb 15;21(4):445-51..
42. Sarzier JS, Evans AJ, Cahill DW. Increased pedicle screw pullout strength with vertebroplasty augmentation in osteoporotic spines. J Neurosurg. 2002 Apr;96(3 Suppl):309-12
43. Rohmiller MT, Schwalm D, Glattes RC, Elalayli TG, Spengler DM. Evaluation of calcium sulfate paste for augmentation of lumbar pedicle screw pullout strength. Spine J. 2002 Jul-Aug;2(4):255-60..
44. Zindrick MR, Wiltse LL, Widell EH, Thomas JC, Holland WR, Field BT, Spencer CW. A biomechanical study of intrapeduncular screw fixation in the lumbosacral spine. Clin Orthop Relat Res. 1986 Feb;(203):99-112..
45. Tsuchiya K, Bridwell KH, Kuklo TR, Lenke LG, Baldus C. Minimum 5-year analysis of L5-S1 fusion using sacropelvic fixation (bilateral S1 and iliac screws) for spinal deformity. Spine (Phila Pa 1976). 2006 Feb 1;31(3):303-8.
46. Dubousset J. Three-dimensional analysis of the scoliotic deformity. In: Weinstein SL, ed. Pediatric Spine: Principles and Practice. New York, NY: Raven Press; 1994
47. Schwab F, Patel A, Ungar B, Farcy JP, Lafage V. Adult spinal deformity-postoperative standing imbalance: how much can you tolerate? An overview of key parameters in assessing alignment and planning corrective surgery. Spine (Phila Pa 1976). 2010 Dec 1;35(25):2224-31.
48. Weinstein SL, Ponseti IV: Curve progression in idiopathic scoliosis. J Bone Joint Surg Am. 1983, 65: 447-455
| How to Cite this Article: Chhabra HS, Yelamarthy PKK. Osteoporosis and its effect on progression of Adult Spinal deformities. International Journal of Spine Apr – June 2016;1(1):15-20 . |

(Abstract) (Full Text HTML) (Download PDF)